Portfolio overview
The Ziccum pipeline of external projects is depicted in a portfolio overview. This gives a general representation of the key steps towards the desired commercialization by entering into license agreements, licensing the LaminarPace technology for specific applications, and the current status of each project. The actual progress in a specific project may proceed via alternative or additional steps, and the timeline varies greatly depending on the resulting read-outs and the counterpart preferences.
Pharmaceutical development in general is subject to very strict confidentiality, and certain collaborations are given without partner name publication, until name disclosure is possible.
The company also pursues earlier dialogues with other counterparts in on-going business development efforts.
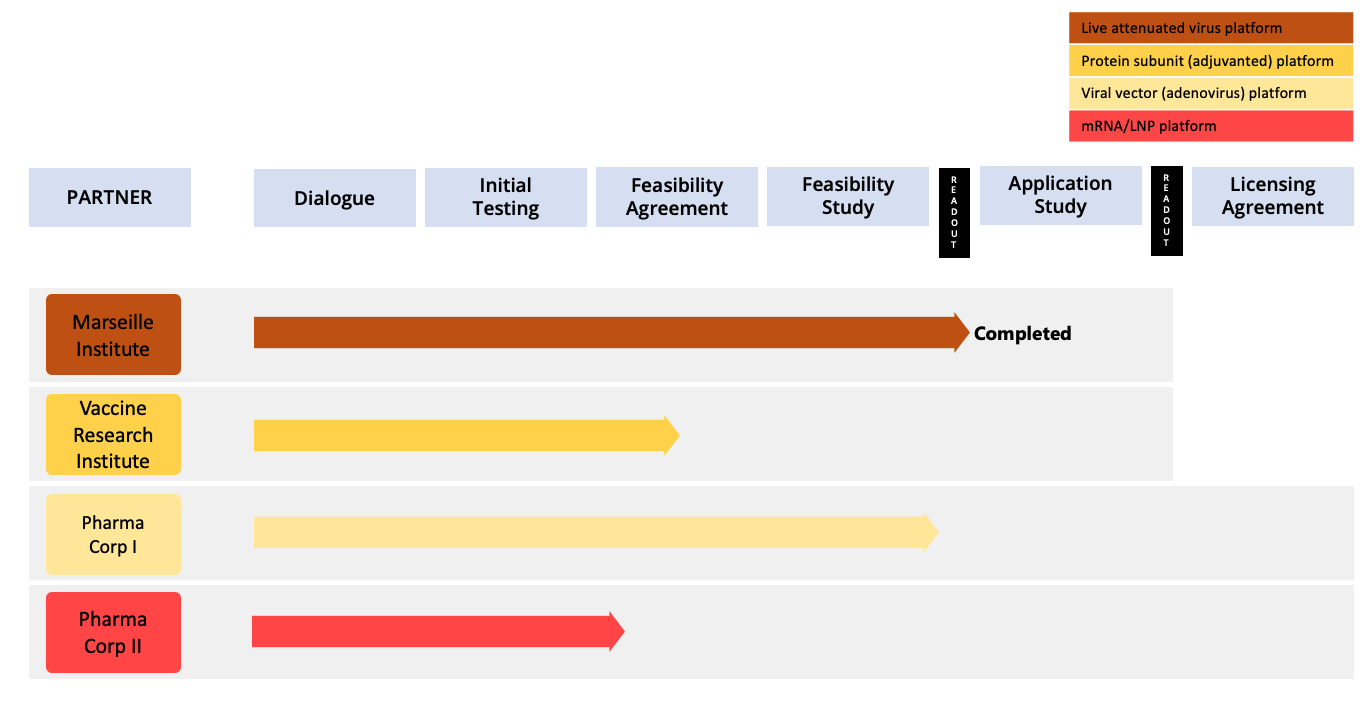
Project portfolio overview as of 30 June, 2022
ZICCUM IN BRIEF
Ziccum's patented technology LaminarPace dries liquid vaccine into a thermostable powder vaccine. Before it is administered, the vaccine powder is mixed with liquid and can then be injected in exactly the same way as a liquid solution vaccine.
The LaminarPace technology is gentle, drying takes place at room temperature, resulting in powder vaccines becoming thermostable. This means that the vaccine can be transported and stored at room temperature, unlike today's vaccines that require costly, complex logistics with an unbroken freeze or cold chain.
confirmed cases of Covid-19 have been reporten to WHO
deaths have been reported to WHO
of a typical vaccine’s carbon footprint comes from its cold chain carbon footprint
of the coverage of HPV vaccines that was achieved in 2019 has been lost. The COVID-19 pandemic fuels the largest setback in childhood vaccinations in three decades.

